16,19,20 This model suggested that the active GPCR conformation opted for by G-protein-coupled receptor kinases (GRKs), arrestins and G proteins is uniform. 16–18 The long-established extended ternary-complex model of GPCR-driven signalling was based on this concept. It was historically assumed that GPCRs exist in two conformations: active and inactive. developed GPCR-ModSim, a web-based portal designed specifically for the homology modelling and MD simulation of GPCRs. 14 Molecular docking is one of the most commonly used methods in GPCR structure-based drug design (SBDD). 13 Multiple-microsecond timescale molecular dynamics (MD) simulations have been used in capturing the process of several drugs binding to β 1- and β 2-adrenergic receptors. 
6 Recently, the Tikhonova group developed a computational protocol which combines concepts from statistical mechanics and cheminformatics to explore the flexibility of the bioamine receptors as well as to identify the geometrical and physicochemical properties which characterise the conformational space of the bioamine family. 6,11,12 Breakthroughs in GPCR crystallography has facilitated novel discovery through virtual screening as well as better off-target rationalisation. 
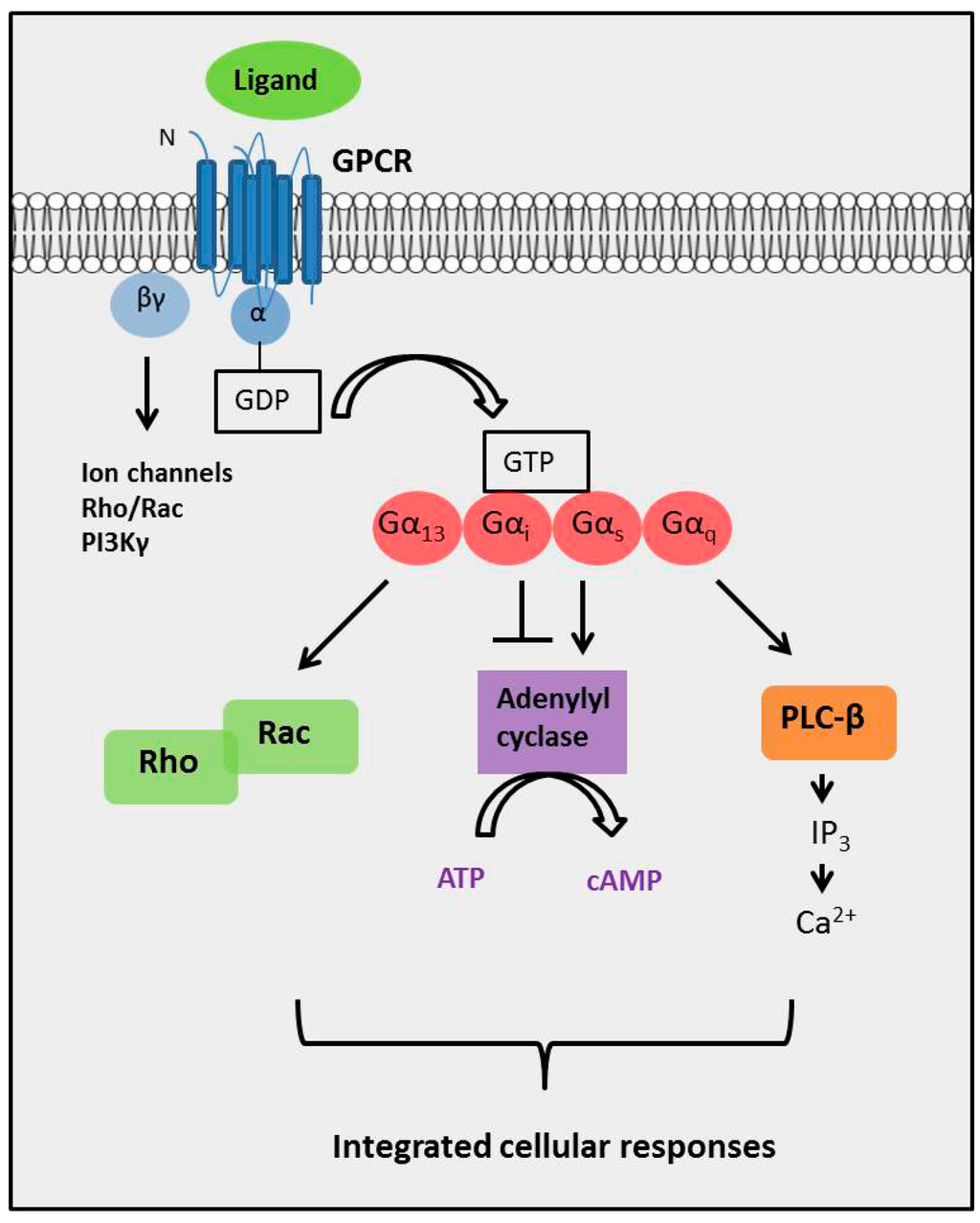
5 Computational biology methods are currently being employed to understand GPCRs as such drug targets. 3 Examples of drugs targeting GPCRs include histamine receptor blockers, opioid agonists, β-blockers and angiotensin receptor blockers. 1,6–10 Hence, there is enormous potential for the development of new drugs targeting these receptors. 2 Currently, approximately thirty to forty percent of marketed pharmaceuticals target GPCRs. 4 The primary function of GPCRs is the transduction of extracellular stimuli into intracellular signals. 4 The conserved structure of GPCRs consists of seven TMD of approximately 25–35 successive amino acid residues that express moderately high levels of hydrophobicity 4 and are characterised by α-helices which span the plasma membrane. 4,5 Their ligands are structurally heterogenous, including natural odorants, nucleotides, amines, peptides, proteins, and lipids. 1–5 GPCRs undertake a plethora of essential physiological functions and are targets for numerous novel drugs. Background The G-protein coupled receptor (GPCR) superfamily consists of structurally similar proteins arranged into families (classes), and is one of the most abundant protein classes in the mammalian genome. The dielectric constant (relative permittivity) of proteins is also discussed in the context of site-specific environmental effects. An overview is provided on family A-C GPCRs their structural differences, GPCR signalling, allosteric binding and cooperativity. Here we provide an introductory and accessible review detailing the computational advances in GPCR pharmacology and drug discovery. GPCRs share some distinctive features, such as the seven transmembrane domains, they also differ in the number of conserved residues in their transmembrane domain. GPCRs take part in many vital physiological functions making them targets for numerous novel drugs. The G-protein coupled receptors (GPCRs) superfamily comprise similar proteins arranged into families or classes thus making it one of the largest in the mammalian genome. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
